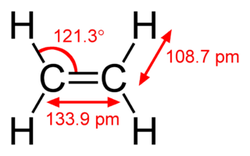
Key Concepts:
Covalent bonds- A bond between two nonmetals
- They usually have prefixes in their name ex. carbon dioxide
- These prefixes show how many of an element are used ex. in the previous ex. the di in dioxide means that 2 oxygens are used
- A pair of electrons are shared
- non conductors
- liquids and gases at room temp
- low melting point
Ionic bonds- A bond between a nonmetal and a metal
- They sometimes contain roman numerals(only when a transition metal is involved) ex. Iron (III) Oxide
- These roman numerals represent the number of valence electrons a transition metal has ex. in the previous ex the (III) means that the iron has three valence electrons
- A pair of electrons are transferred
- rigid, crystalline solids
- high melting point
- usually solid at room temp
- conducts in molten (liquid) state
- mostly soluble in water
Metallic bonds- A bond between two metals
- sea of electrons (mobile valence electrons)
- Malleable, ductile, conducts, not strong
Covalent bonds- A bond between two nonmetals
- They usually have prefixes in their name ex. carbon dioxide
- These prefixes show how many of an element are used ex. in the previous ex. the di in dioxide means that 2 oxygens are used
- A pair of electrons are shared
- non conductors
- liquids and gases at room temp
- low melting point
Ionic bonds- A bond between a nonmetal and a metal
- They sometimes contain roman numerals(only when a transition metal is involved) ex. Iron (III) Oxide
- These roman numerals represent the number of valence electrons a transition metal has ex. in the previous ex the (III) means that the iron has three valence electrons
- A pair of electrons are transferred
- rigid, crystalline solids
- high melting point
- usually solid at room temp
- conducts in molten (liquid) state
- mostly soluble in water
Metallic bonds- A bond between two metals
- sea of electrons (mobile valence electrons)
- Malleable, ductile, conducts, not strong
Ionic, Covalent, and Metallic Bonds. Dir. Khan Acadamy. Perf. Khan Acadamy. YouTube. N.p., n.d. Web. 29 May 2014.
Lewis symbols- Shows an element and the number of valence electrons it has
- valence electrons are represented by dots. The dots are arranged one by one on the top, bottom, and both sides
- Each side must have one dot before it can have two
- valence electrons are represented by dots. The dots are arranged one by one on the top, bottom, and both sides
- Each side must have one dot before it can have two
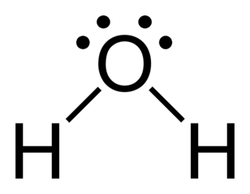 Wikipedia. Water 2D Flat. Digital image. Wikipedia. N.p., n.d. Web. 29 May 2014.
Wikipedia. Water 2D Flat. Digital image. Wikipedia. N.p., n.d. Web. 29 May 2014.
Lewis structures- are diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons
- Lewis structures show each atom and its position in the
structure of the molecule using its chemical symbol. Lines are drawn between atoms that are bonded to one another (pairs of dots can be used instead of lines). - Excess electrons that form lone pairs are represented as pairs of dots, and are placed next to the atoms.
- Lewis structures show each atom and its position in the
structure of the molecule using its chemical symbol. Lines are drawn between atoms that are bonded to one another (pairs of dots can be used instead of lines). - Excess electrons that form lone pairs are represented as pairs of dots, and are placed next to the atoms.
The octet rule and its exceptions
Octet Rule Exceptions. Dir. JCCCEM141. Perf. JCCCEM141. YouTube. N.p., n.d. Web. 29 May 2014.
Electronegativity- energy needed to take an electron
Electronegativity - Bonding. Dir. Chemistry Lectures. Perf. Chemistry Lectures.YouTube. N.p., n.d. Web. 29 May 2014.